|
·
UltraSep ES POLYOL,
optically active as
a) UltraSep ES AMINOPOLYOL H
(Aminodesoxyhexitol)
|
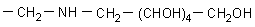
|
|
|
b) UltraSep ES AMINOPOLYOL G
(Aminodesoxy-d-glucose) |
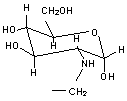 |
|
| · UltraSep ES
AMINOTETRAOL |
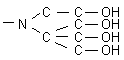 |
|
| Alcoholic
hydroxyls and amino groups on the surface of this packing are responsible
for a blocking of residual silanols. This means, that polar compounds can be
excellently separated by normal phase chromatography. Example:
Stereoisomeric organic N-oxides.
AMINOTETRAOL
is
a very versatile phase. In the pharmaceutical industry it is advantageously
used also with aqueous and aqueous-organic eluents. One can use the phase to
separate sugars and more. Furthermore, the packing works as a weak ion
exchanger and separates with weak eluents, e. g., organic carbon acids at pH
3. |
·
UltraSep
ES DTA (= Diethylenetriamine)
This packing is designated for special applications with amino packings. To
this group belongs also UltraSep ES TTA |
– CH2 – NH – CH2 –
CH2
– NH – CH2 – CH2 – NH2
|
|
·
UltraSep
ES Tetraminool (TTA)
(= Triethylenetetramine)
This packing has in comparison with DTA a doubled spacer chain and three
ethylene residues connected by nitrogen atoms. Beside the primary amino
group at the end of the chain there exists an alcoholic hydroxyl group at the
spacer. |
– CH2 – NH – (CH2 – CH2
– NH)2 – CH2 – CH2 – NH2
|
|
·
UltraSep ES RP8F (Fluorooctylsilica)
This packing is a fluorooctylsilica, where the H-atoms of each C-atom are
substituted by 17 fluorine atoms. This packing is an alternative RP phase
with relatively rigid strong hydrophobic chains for special applications. |
– (CF2)7 – CF3 |
|
·
UltraSep ES PEO (Polyethyleneglycolsilica)
UltraSep ES PEO is a packing on the basis of polyethyleneoxide chains.
Within the chain C2 segments alternate with oxygen atoms. In total there
are 26 atoms within the chain, 18 of them are carbon atoms as it is the
case in standard reversed phases. Their properties are levelled by the
oxygen atoms and combined with interactions due to hydrogen bridges (OH....O;
NH....O). Therefore the PEO packing can be used both for
reversed phase chromatography and for normal phase chromatography (see
application PEO1 and PEO2,
respectively). With adequate compounds it exhibits specific selectivity
effects as well as shorter retention times in comparison to RP18 materials. |
– (CH2 – CH2 – O)6-9 – CH2 –
CH2 – OCH3
Application PEO1
Application PEO2
|
|
·
UltraSep ES PFP
UltraSep ES PFP is a pentafluorophenylalkylsilica of the RP8 reversed
phase type with outstanding properties. On average it yields the
retention times of the RP18 packing type with the following
characteristics:
 | Compounds which are not separated enough by
normal RP18 phases, potentially can be resolved sufficiently using the PFP packing (purity screening). |
 | PFP is very attractive for pharmaceutically
relevant substances, especially for bases, which in principle
show a small tailing (application PFP1 and PFP2).
Base pairs which are not separated by normal RP packings, can be
resolved (application PFP3). |
 | PFP packings behave significantly different in
comparison to normal RP packings. For instance in the test mixture of
toluene, propyl benzene and pentyl benzene with rising water content
the peaks become slender. At a composition of the acetonitrile/water
mixture of 1/1 125,000 theoretical plates per m are accomplished
already with 4 µm material. |
 | PFP phases together with the normal RP phases and
RP amid phases or also with PEO phases yield an advantageous "selectivity
trio" and "selectivity quartet", respectively. |
|
Application PFP1
Application PFP2
Application PFP3
|
|
·
UltraSep ES C30 (RP-packing)
With long C30 chains normal material (pore diameter of 120 Å) as well as
material with large pores (pore diameter of 300 Å = D300) are offered. |
– (CH2)29 – CH3
Application
|
|
·
UltraSep ES ADC (= Aminodicarboxy)
As active group aminodiacetic acid was bound over a spacer with 6 C atoms. |
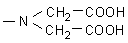 |
|
·
UltraSep
ES TC (=
Triolcarboxy)
In this packing a carboxylic group was combined with a hydroxymethyl
cluster having the basic ligand structure shown in the formula. |
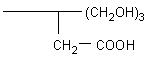 |
|
·
UltraSep
ES C12 TA (= Triamino)
This
packing is strongly polar and possesses branched aliphatic chains
containing two terminal primary amino groups and one secondary amino
group. |
|
|
·
UltraSep
ES C10 w-COOH
This is a medium polar packing with an unbranched aliphatic
carbon chain and two carboxylic groups, one is terminal . It shows typical RP
behaviour. |
|
|
·
UltraSep ES AMID RP18 500
This packing is a lowly charged methyloctadecylsilica gel with a specific
selectivity.
|
|
|
|
Shield - Carrier (Embedded
Groups) — RP packings with polar selectivity
[see GIT 46 (2002),
number 1 . Please ask for a reprint.]
suitable
 |
for purely aqueous media ("Aq")
without phase collaps |
 |
for the separation of highly
polar
compounds |
|
·
UltraSep ES AMID RP18A
3-(Octadecanoylamino)propylslilyl-Silica. The packing carries an amide
group between spacer and RP chain (so called Embedded
Group). Spec. area 280 m²/g. |
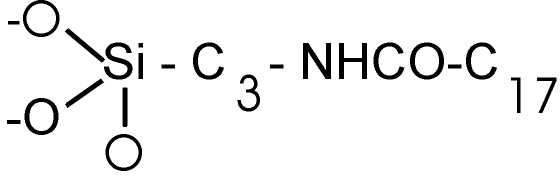
Application
|
|
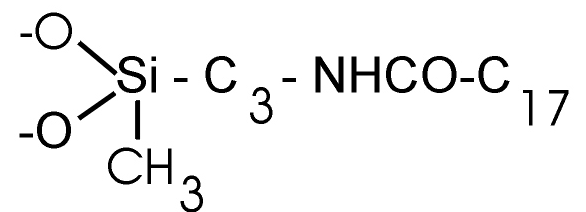
·
UltraSep ES AMID RP18AM
The packing possesses additionally a methyl group at the silyl residue. Spec.
area 420 m²/g. |
|
|
·
UltraSep ES AMID H RP18PA
3-(Heptadecylureido)-propylslilyl-Silica. The packing carries a urea group
instead of the amide group.
|
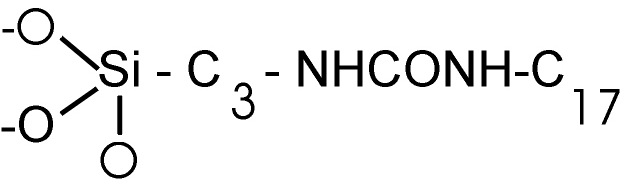
Application1
Application2
|
|
|
·
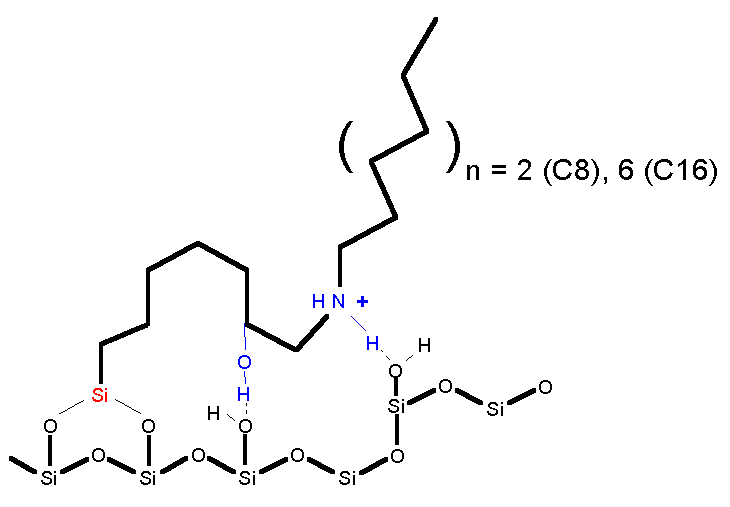
UltraSep ES CHAIR
UltraSep
ES CHAIR
is a special phase obtained by chemical bonding of a sufficiently long non
polar chain to the silica. The chain bears one or two polar substituents at
the first third or at the middle part.
The
groups show a pronounced interaction with silanols. Therefore, depending on
the chromatographic conditions for a unique chain folding, a chair like
configuration can be expected ( figure).
It
is well known, that normal phases with polar groups show RP-behaviour
too. With chair phases, that dual behaviour was intentional, so to speak,
typified. In this way we can synthesize tailor-made selectivities, changing
the length of the non polar chain R or
the nature and/or number (1 or 2) of the polar groups.
UltraSep
ES CHAIR is often used, e. g. in the pharmaceutical industry and in
biochemical institutions. Example: Cephalosporines.
|
|
UltraSep
ES CHAIR structure (on principle)
|
|
P
= Polar substituent (interaction with silanoles)
R = Alkyl chain with 2 – 12 carbon
atoms
|
Application |
|
·
UltraSep ES UREIDO
Ureidopropylsilyl-silica. Normal phase
with a urea group at the end of the propyl spacer |
|
|
|
|
